
Pascal THEROND
Signal transduction and control of morphogenesis in Drosophila
Main interests
- Cell-to-cell interactions
- Signaling molecules as organizers in animal development
- Release and extra-cellular transportation of morphogens
- Tissue morphogenesis and basement membrane
- Homeostasis of glial and neuronal cells during aging
Scientific Questions
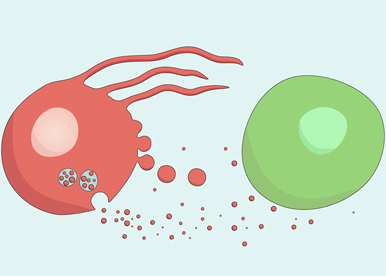
Cell-to-cell interactions are universal processes required during embryonic development and adult life in all metazoans. Although fundamental, the cellular mechanisms necessary for these processes remain poorly understood. How conserved secreted ligands such as morphogens function as organizers in animal development, and how gene expressions is controlled by these signaling molecules and translated into cell behavior are fundamental questions that our research program aims to explore. Our studies are focussed on the Hedgehog (Hh), Decapentaplegic (Dpp) and Wingless (Wg) secreted morphogens and employ Drosophila melanogaster as a model for similar processes in vertebrates that are less accessible for genetic study. We also study the protective role of morphogens in adult life and the deleterious effects of morphogen signaling in human tumors. We hope to obtain a comprehensive understanding of morphogen activity and to identify conserved regulators of cell to cell interactions.
Our Strategy
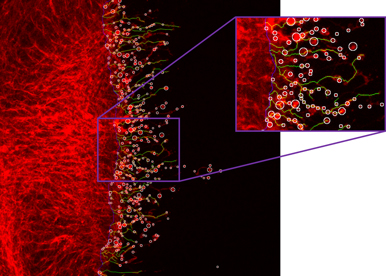
We are developing an exciting research framework to investigate the spatio-temporal dynamics of morphogen gradient combining cutting-edge genetic, molecular biology, biochemical, imaging and electronic microscopy methodologies. (1) Our work showed that the Hh morphogenetic gradient is composed of several pools; using two epithelia we are currently evaluating the individual contribution of the different extra-cellular Hh carriers which have been proposed to sustain Hh spreading (lipoprotein particles, filopodia, multimers and extra-cellular vesicles). (2) We identified a protein complex which governs access to the nucleus of the Gli transcription factor that mediates Hh signal. Tools such as mass spectrometry, phospho-specific antibody, bimolecular complementation of fluorescence (BiFC), have been developed to reach a comprehensive understanding of the molecular interaction present in this protein complex at different level of activation. (3) Since tissue morphogenesis and basal membrane modulation must be coupled, we will use our knowledge of morphogen regulation to investigate how wing disc patterning and the dynamics of basement membrane structure are coupled. (4) Finally, we are using the adult fly to analyze the function of Hh in adult glial cells. We therefore developed a new exploratory project investigating the role of Hh in Drosophila adult brain and in human glioblastoma.
Research Aims

Release and transportation of Hh molecules
The goal of this project is to understand how Hh is secreted. By developing an interface between computational science and biology, we are analysing the spatiotemporal dynamics of Hh movement in live Drosophila epithelia with single molecule tracing. The combination of genetic manipulations to modify secretion of Hh with ultra-structure analysis will allow us to determine the contribution of the known Hh carriers in the establishment of the Hh gradient.
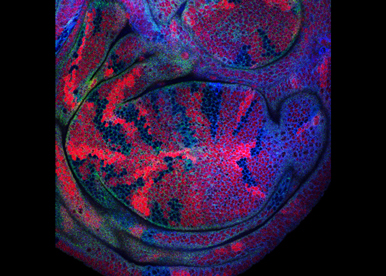
Morphogen Signaling
We intend to identify conformational changes in the proteins of the Hh signaling complex. Multiple protein associations have been quantified and visualized in vivo. We aim to clarify the changes in binding association, and describe the protein regions involved in the switch of conformations at different levels of activation. This will improve our understanding of how the morphogenetic gradient of extra-cellular information is converted into a gradient of signaling activity.
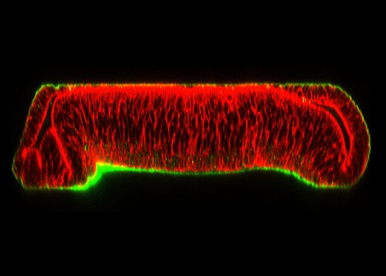
Coupling of Tissue Patterning and Basal Membrane (BM) Dynamics
We aim to determine in vivo whether the BM controls morphogen signaling, and in turn, whether morphogens affect BM dynamics. We are exploring the role of Perlecan (Pcan), a central component of the BM, known to maintain the BM’s organization. We will evaluate the consequence of Pcan depletion on morphogen signaling. Because Pcan expression is dynamic in the wing disc, we aim to identify the morphogen regulating pcan transcription.
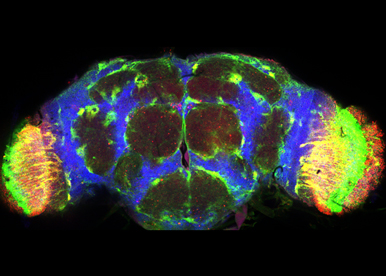
Hh signaling in neuro-protection
We aim to explore the role of the Hh signaling in adult brain. Overall our data suggest that Hh signaling in glial cells participates in protecting against the aberrant misfolding of proteins during the ageing process and in neurodegenerative disease models. We will decipher the function of Hh in the brain, exploring the role of the Hh signaling in glial and neuronal cell homeostasis and identifying regulators that participate in the Hh neuroprotective effect.
Selected Publications
- Hedgehog Elicits Signal Transduction by Means of a Large Complex Containing the Kinesin Related Protein Costal-2. Robbins, D., Nybbacken, K., Kobayashi, R., Sisson, J., Bishop, J. M., and Thérond. Cell (1997) vol. 90: 225-234. doi: 10.1016/s0092-8674(00)80331-1.
- Cholesterol Modification of Hedgehog Is Required for Functional Trafficking and Movement, Revealing an Asymmetric Cellular Response to Hedgehog. Gallet A., Rodriguez R., Ruel L., and PP. Thérond. Developmental Cell (2003) vol. 4, 191-204. doi: 10.1016/s1534-5807(03)00031-5. Cited in Faculty of 1000 Biology.
- Stability and Association of Smoothened, Costal2, and Fused with Cubitus-interruptus Is Regulated by Hedgehog. Ruel L., Rodriguez R., Gallet A., Lavenant-Staccini L., and P P. Thérond. Nature Cell Biology (2003) vol. 5: 907-913. doi: 10.1038/ncb1052. Reviewed in Current Biology, vol. 14, R67-69, 2004. Cited in Faculty of 1000 Biology.
- Cellular trafficking of the glypican Dally-like is required for full-strength Hedgehog signaling and wingless transcytosis. Gallet A, Staccini-Lavenant L, Thérond PP. Developmental Cell. 2008 May;14(5):712-25. doi: 10.1016/j.devcel.2008.03.001.PMID:18477454
- The Long-Range Activity of Hedgehog Is Regulated In the Apical Extracellular Space by the Glypican Dally and Hydrolase Notum. Ayers K., Gallet A., Staccini L., and PP. Thérond. Developmental Cell, (2010), 18, 605–620. doi: 10.1016/j.devcel.2010.02.015.
- Distinct phosphorylations on kinesin costal-2 mediate differential hedgehog signaling strength. Ranieri N, Ruel L, Gallet A, Raisin S, Thérond PP. Developmental Cell, 2012 Feb 14;22(2):279-94. doi:10.1016/j.devcel.2011.12.002. Epub 2012 Feb 2.PMID: 22306085
- Switch of PKA substrates from Cubitus interruptus to Smoothened in the Hedgehog signalosome complex. Ranieri N, Laurent Ruel* and Pascal Thérond*. Nature Communication. 2014 Oct 7;5:5034. doi: 10.1038/ncomms6034.
- The ESCRT Machinery Regulates the Secretion and Long-Range Activity of Hedgehog. Tamas Matusek, Franz Wendler, Sophie. Polès, Sandrine Pizette, Gisela D’Angelo, Maximilian Fürthauer and Pascal P. Thérond. Nature (2014) Dec 4;516(7529): 99-103. doi: 10.1038/nature13847.
- Endocytosis of Hedgehog through dispatched regulates long-range signaling. D'Angelo G,Matusek T, Pizette S, Thérond PP. Developmental Cell. 2015 Feb 9;32(3):290-303. doi:10.1016/j.devcel.2014.12.004. Epub 2015 Jan 22.PMID: 25619925
- Hedgehog Signaling Modulates Glial Proteostasis and Lifespan. Rallis A, Navarro JA, Rass M, Hu A, Birman S, Schneuwly S, Thérond PP. Cell Reports (2020) Feb 25;30(8):2627-2643.e5. doi: 10.1016/j.celrep.2020.02.006.
Hedgehog Links
Researchers
 RUEL Laurent - +33 489150758
RUEL Laurent - +33 489150758 PIZETTE Sandrine - +33 489150763
PIZETTE Sandrine - +33 489150763 MATUSEK Tamas - +33 489150763
MATUSEK Tamas - +33 489150763
Engineers & Technicians
 LAVENANT-STACCINI Laurence - +33 489150763
LAVENANT-STACCINI Laurence - +33 489150763
Students
 BELADOUI Amira - +33 489150763
BELADOUI Amira - +33 489150763
Masters
 TCHENAR Jihan - +33 489150763
TCHENAR Jihan - +33 489150763
- Bare, Y, Matusek, T, Vriz, S, Deffieu, MS, Thérond, PP, Gaudin, R et al.. TMED10 mediates the loading of neosynthesised Sonic Hedgehog in COPII vesicles for efficient secretion and signalling. Cell Mol Life Sci. 2023;80 (9):266. doi: 10.1007/s00018-023-04918-1. PubMed PMID:37624561 PubMed Central PMC11072717.
- Bonche, R, Smolen, P, Chessel, A, Boisivon, S, Pisano, S, Voigt, A et al.. Regulation of the collagen IV network by the basement membrane protein perlecan is crucial for squamous epithelial cell morphogenesis and organ architecture. Matrix Biol. 2022;114 :35-66. doi: 10.1016/j.matbio.2022.10.004. PubMed PMID:36343860 .
- Hurbain, I, Macé, AS, Romao, M, Prince, E, Sengmanivong, L, Ruel, L et al.. Microvilli-derived extracellular vesicles carry Hedgehog morphogenic signals for Drosophila wing imaginal disc development. Curr Biol. 2022;32 (2):361-373.e6. doi: 10.1016/j.cub.2021.11.023. PubMed PMID:34890558 .
- Marcetteau, J, Matusek, T, Luton, F, Thérond, PP. Arf6 is necessary for senseless expression in response to wingless signalling during Drosophila wing development. Biol Open. 2021;10 (12):. doi: 10.1242/bio.058892. PubMed PMID:34779478 PubMed Central PMC8656404.
- Pizette, S, Matusek, T, Herpers, B, Thérond, PP, Rabouille, C. Hherisomes, Hedgehog specialized recycling endosomes, are required for high level Hedgehog signaling and tissue growth. J Cell Sci. 2021;134 (10):. doi: 10.1242/jcs.258603. PubMed PMID:34028543 .
- Gore, T, Matusek, T, D'Angelo, G, Giordano, C, Tognacci, T, Lavenant-Staccini, L et al.. The GTPase Rab8 differentially controls the long- and short-range activity of the Hedgehog morphogen gradient by regulating Hedgehog apico-basal distribution. Development. 2021;148 (5):. doi: 10.1242/dev.191791. PubMed PMID:33547132 .
- Prince, E, Marcetteau, J, Thérond, PP. Circulating Hedgehog: a fresh view of a classic morphogen. Development. 2020;147 (24):. doi: 10.1242/dev.186395. PubMed PMID:33355241 .
- Bonche, R, Chessel, A, Boisivon, S, Smolen, P, Thérond, P, Pizette, S et al.. Two different sources of Perlecan cooperate for its function in the basement membrane of the Drosophila wing imaginal disc. Dev Dyn. 2021;250 (4):542-561. doi: 10.1002/dvdy.274. PubMed PMID:33269518 .
- Matusek, T, Marcetteau, J, Thérond, PP. Functions of Wnt and Hedgehog-containing extracellular vesicles in development and disease. J Cell Sci. 2020;133 (18):. doi: 10.1242/jcs.209742. PubMed PMID:32989011 .
- Rallis, A, Navarro, JA, Rass, M, Hu, A, Birman, S, Schneuwly, S et al.. Hedgehog Signaling Modulates Glial Proteostasis and Lifespan. Cell Rep. 2020;30 (8):2627-2643.e5. doi: 10.1016/j.celrep.2020.02.006. PubMed PMID:32101741 .
- Matusek, T, Thérond, P, Fürthauer, M. Functional Analysis of ESCRT-Positive Extracellular Vesicles in the Drosophila Wing Imaginal Disc. Methods Mol Biol. 2019;1998 :31-47. doi: 10.1007/978-1-4939-9492-2_3. PubMed PMID:31250292 .
- Giordano, C, Ruel, L, Poux, C, Therond, P. Protein association changes in the Hedgehog signaling complex mediate differential signaling strength. Development. 2018;145 (24):. doi: 10.1242/dev.166850. PubMed PMID:30541874 .
- D'Angelo, G, Matusek, T, Pizette, S, Thérond, PP. Endocytosis of Hedgehog through dispatched regulates long-range signaling. Dev Cell. 2015;32 (3):290-303. doi: 10.1016/j.devcel.2014.12.004. PubMed PMID:25619925 .
- Matusek, T, Wendler, F, Polès, S, Pizette, S, D'Angelo, G, Fürthauer, M et al.. The ESCRT machinery regulates the secretion and long-range activity of Hedgehog. Nature. 2014;516 (7529):99-103. doi: 10.1038/nature13847. PubMed PMID:25471885 .
- Ranieri, N, Thérond, PP, Ruel, L. Switch of PKA substrates from Cubitus interruptus to Smoothened in the Hedgehog signalosome complex. Nat Commun. 2014;5 :5034. doi: 10.1038/ncomms6034. PubMed PMID:25289679 .
- Briscoe, J, Thérond, PP. The mechanisms of Hedgehog signalling and its roles in development and disease. Nat Rev Mol Cell Biol. 2013;14 (7):416-29. doi: 10.1038/nrm3598. PubMed PMID:23719536 .
- Ayers, KL, Mteirek, R, Cervantes, A, Lavenant-Staccini, L, Thérond, PP, Gallet, A et al.. Dally and Notum regulate the switch between low and high level Hedgehog pathway signalling. Development. 2012;139 (17):3168-79. doi: 10.1242/dev.078402. PubMed PMID:22872085 .
- Aikin, R, Cervantes, A, D'Angelo, G, Ruel, L, Lacas-Gervais, S, Schaub, S et al.. A genome-wide RNAi screen identifies regulators of cholesterol-modified hedgehog secretion in Drosophila. PLoS One. 2012;7 (3):e33665. doi: 10.1371/journal.pone.0033665. PubMed PMID:22432040 PubMed Central PMC3303847.
- Thérond, PP. Release and transportation of Hedgehog molecules. Curr Opin Cell Biol. 2012;24 (2):173-80. doi: 10.1016/j.ceb.2012.02.001. PubMed PMID:22366329 .
- Ranieri, N, Ruel, L, Gallet, A, Raisin, S, Thérond, PP. Distinct phosphorylations on kinesin costal-2 mediate differential hedgehog signaling strength. Dev Cell. 2012;22 (2):279-94. doi: 10.1016/j.devcel.2011.12.002. PubMed PMID:22306085 .
2023 - Prix Eloi Collery - Académie Nationale de Médecine
2015 - Prize of Excellence - Université Côte d'Azur UCA
2015 - Gallet and Breton Prize from the French Academy of Medicine
2013 - Excellence Award (Prime d'excellence scientifique) – CNRS
2009 - Excellence Award (Prime d'excellence scientifique) – CNRS
2009 - Victor Noury Prize from the French Academy of Science
2004 - Marie Curie Host for Research Training
2001 - Young Investigator Program award - EMBO
1998 - ATIP Program in Cell Biology - CNRS
1992 - G.W. Hooper Foundation Fellowship

Pascal THÉROND récipiendaire du prix Eloi COLLERY 2023 de l’Académie Nationale de Médecine
Read More

Hedgehog signalling modulates glial proteostasis and lifespan
Read More

UCA annual Award Ceremony: 7 iBV members recognised for their scientific contributions
Read More
iBV - Institut de Biologie Valrose
"Centre de Biochimie"
Université Nice Sophia Antipolis
Faculté des Sciences
Parc Valrose
06108 Nice cedex 2
