
Franck DELAUNAY
Chronobiology
Main interests
- Circadian clock
- Metabolism
- Cell cycle
- Systems biology
Scientific Questions

The mammalian circadian timing system coordinates a variety of molecular, cellular and physiological processes over the 24 h in synchrony with the light/dark cycle. The fundamental mechanism underlying circadian rhythms is a molecular clock present in virtually all body cells. The system is organized hierarchically with a central clock in the hypothalamus that coordinates peripheral clocks through internal synchronizers. Misalignment or disruption of the human circadian timing system has been linked to pathologies including metabolic disorders, sleep phase syndromes and cancer. Although we currently have a comprehensive view of how core clock genes work and interact together to generate oscillations, we still do not fully understand how they crosstalk with other essential cellular mechanisms. In that context, we study how mammalian peripheral circadian clocks interact with the cell cycle and metabolic processes and determine the relevance of these interactions in health and disease.
Our Strategy
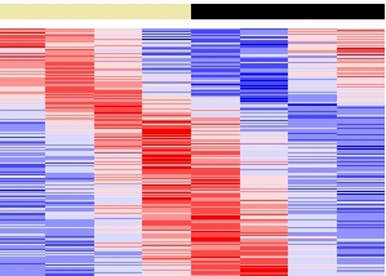
To study the interaction between the circadian clock and metabolism, we use a panel of tools and approaches including systemic and conditional knockout mouse models, culture of primary cells, functional genomics, nutritional challenges and, monitoring of circadian physiology and behavior. Our second project on the interaction between the circadian clock and the cell cycle involves mainly a systems biology approach that combines multispectral fluorescent reporter systems, high throughput time lapse imaging, bioinformatics and mathematical modelling in collaboration with mathematicians and computer scientists.
Research Aims
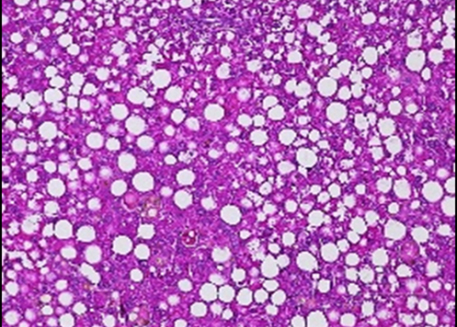
We have established that Krüppel Like Factor 10 (KLF10) is a clock-controlled transcription factor in liver. Our goal is to precisely decipher its role in the circadian coordination of key metabolic processes in hepatocytes. We also aim to evaluate its relevance in the context of non-alcoholic fatty liver disease which is rising worldwide.
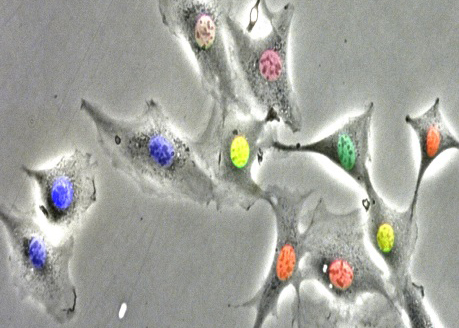
We have demonstrated that the two oscillators driving progression of the cell cycle and the circadian clock respectively are robustly coupled through a phase locking mechanism in unperturbed dividing mammalian cells. Our main goal is to understand the dynamics of this coupling and determine how this phenomenon might be exploited to design chronotherapeutics approaches for cancer treatment.
Researchers
 GRECHEZ-CASSIAU Aline - +33 489150826
GRECHEZ-CASSIAU Aline - +33 489150826 TEBOUL Michèle - +33 489150825
TEBOUL Michèle - +33 489150825 SATNEY Isabelle - +33 489153704
SATNEY Isabelle - +33 489153704 SIBILLE Brigitte - +33 489153704
SIBILLE Brigitte - +33 489153704 ROUSSEAU Anne-Sophie - +33 489153704
ROUSSEAU Anne-Sophie - +33 489153704
PreDocs
 MERCIER Julien - +33 489153704
MERCIER Julien - +33 489153704 MARCHANDISE-FRANQUET Sophia - +33 489153704
MARCHANDISE-FRANQUET Sophia - +33 489153704
Engineers & Technicians
 GUéRIN Sophie - +33 489150827
GUéRIN Sophie - +33 489150827 TAHRAOUI Sana - +33 489153704
TAHRAOUI Sana - +33 489153704
Masters
 SA Alexandre - +33 R
SA Alexandre - +33 R
iBV - Institut de Biologie Valrose
"Sciences Naturelles"
Université Nice Sophia Antipolis
Faculté des Sciences
Parc Valrose
06108 Nice cedex 2
