
Marie-Christine CHABOISSIER
Sex determination and fertility
Main interests
- Sex determination in mammals
- Normal and pathogenic development of the gonad
- Ovarian homeostasis and repair
- WNT/b-catenin signalling and Sox genes
Scientific Questions
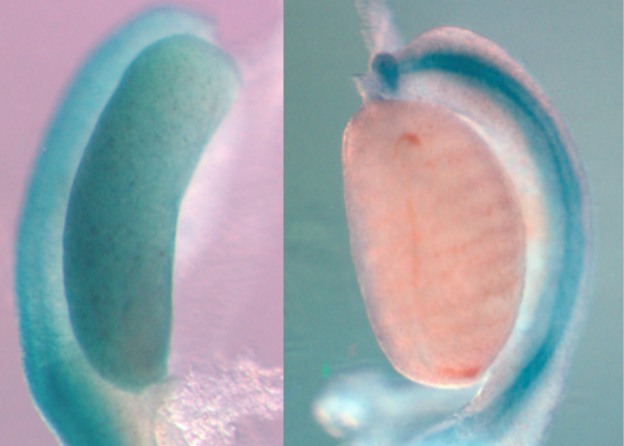
In mammals, the pivotal decision to develop as male or female begins with the differentiation of the bipotential gonad allowing the development of two highly specialized organs: the testis or the ovary. This will condition the whole sexual fate of the individual. At a molecular level, sex determination depends on two antagonistic molecular pathways. The SRY gene located on the Y chromosome initiates testicular development by up-regulating the transcription factor SOX9 in XY embryos. In XX embryos, the stabilization of ovarian differentiation is secured by the WNT/b-catenin signalling pathway mediated by R-spondin1 and by the transcription factor, FOXL2. Our lab has just shown that one isoform of WT1 is essential to initiate ovarian development as described in https://www.science.org/stoken/author-tokens/ST-1527/full . However, the knowledge on mechanisms governing sex determination is still fragmented and consequently about 50% of gonadal dysgenesis remains unexplained in humans.
Our Strategy
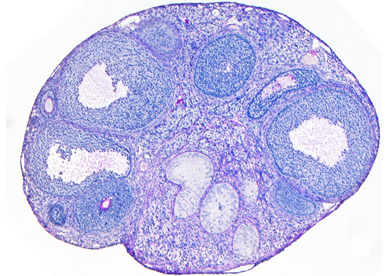
Understanding sex determination or how two completely different organs, the testis or the ovary, differentiate from the same precursor is a unique quest in biology. We have a long-standing interest in the processes driving sex determination, sexual differentiation, and gonad homeostasis. We have shown that Rspo1 promotes progenitor cell proliferation in the bipotential gonad. After their ingression into the gonads, cells in the XY gonads stop expressing Rspo1 and activate the testis determining genes Sry and Sox9. We have shown that Sox9 is required for the differentiation of Sertoli cells, the supporting cells of the testis and in turn male development. In XX gonads, the progenitor cells maintain Rspo1 expression after entering the gonads. We have identified that -KTS isoform of WT1 is the long-sought ovarian determinant and induces the differentiation of the progenitor cells into granulosa cells, the somatic cells that will form the ovarian follicles after birth. Next, Rspo1 is required for embryonic granulosa cell maintenance during embryogenesis. Mutations in these key factors triggers gonadal dysgenesis ranging from streak gonad to sex reversal. To address these questions, we combine genetic, molecular and genomic approaches.
Research Aims
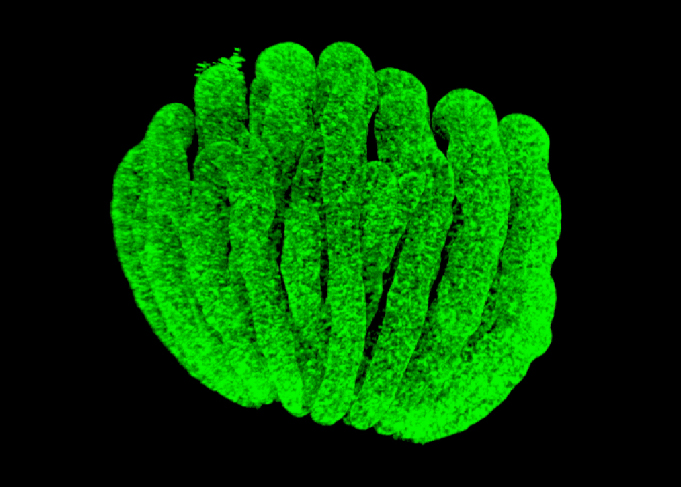
Testis development is determined by the activation of SRY and SOX9. Around the same time, the down-regulation of RSPO1/WNT/b-catenin signalling is critical for the differentiation of this organ. There are still many questions that need to be answered in order to understand testis development, such as why a limited number of cells become supporting cells or steroidogenic cells, how RSPO1/WNT/b-catenin signalling is down-regulated, etc.
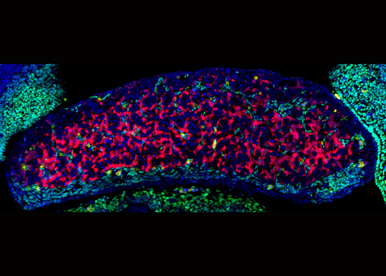
In XX gonads, the -KTS isoform of WT1 induces ovarian differentiation and Rspo1 is necessary to sustain the ovarian fate, but there are still fundamental questions to be answered, such as how the cell populations of the ovary are specified, how RSPO1 protects the ovary from reprogramming as ovotestis, etc. Understanding the normal role of this pathway is a prerequisite for the study of ovarian pathogenesis.
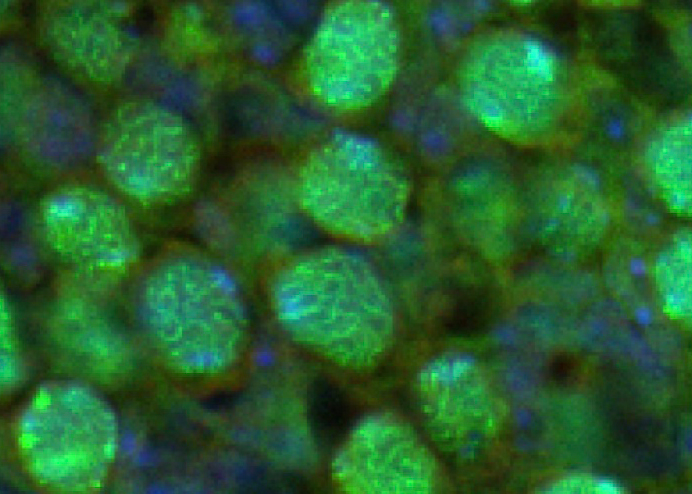
The gonad is the site of germ cells differentiation. It is now clear that the fate of the gonad and germ cells are closely linked and the exact cellular and molecular mechanism governing sex-specific germ cell differentiation remain to be elucidated.
Our team is dedicated to trying to answer some of these questions. These findings will lead to better understanding of some of the many cases of gonadal dysgenesis and cancers that have yet to find an explanation.
Researchers
 GILLOT Isabelle - +33 489150727
GILLOT Isabelle - +33 489150727 DE CIAN Marie-Cécile - +33 489150726
DE CIAN Marie-Cécile - +33 489150726 PEREA-GOMEZ Aitana - +33 489150726
PEREA-GOMEZ Aitana - +33 489150726 DHELLEMMES Magali - +33 489150728
DHELLEMMES Magali - +33 489150728
PreDocs
 DUBOS Lauryane - +33 489150726
DUBOS Lauryane - +33 489150726
Engineers & Technicians
 GRéGOIRE Elodie - +33 489150726
GRéGOIRE Elodie - +33 489150726 ROZIER Agathe - +33 489150726
ROZIER Agathe - +33 489150726
Students
 TELIOU Konstantina - +33 489150726
TELIOU Konstantina - +33 489150726
Recent publications
- Djari, C, Mayère, C, Guy, M, Perea-Gomez, A, Barreau, P, Rozier, A et al.. Ovarian development is driven by early spatiotemporal priming of the coelomic epithelium. bioRxiv. 2025; :. doi: 10.1101/2025.09.24.678234. PubMed PMID:41040240 PubMed Central PMC12485839.
- Perea-Gomez, A, Bellido Carreras, N, Dhellemmes, M, Tang, F, Le Gallo, C, Chaboissier, MC et al.. NR2F2 is required in the embryonic testis for fetal Leydig cell development. Elife. 2025;14 :. doi: 10.7554/eLife.103783. PubMed PMID:40637239 PubMed Central PMC12245173.
- Wilhelm, D, Perea-Gomez, A, Newton, A, Chaboissier, MC. Gonadal sex determination in vertebrates: rethinking established mechanisms. Development. 2025;152 (6):. doi: 10.1242/dev.204592. PubMed PMID:40162719 .
- Grégoire, É, De Cian, MC, Detti, M, Gillot, I, Perea-Gomez, A, Chaboissier, MC et al.. [Sex determination, it is all about timing]. Med Sci (Paris). 2024;40 (8-9):627-633. doi: 10.1051/medsci/2024095. PubMed PMID:39303114 .
- Gregoire, EP, De Cian, MC, Migale, R, Perea-Gomez, A, Schaub, S, Bellido-Carreras, N et al.. The -KTS splice variant of WT1 is essential for ovarian determination in mice. Science. 2023;382 (6670):600-606. doi: 10.1126/science.add8831. PubMed PMID:37917714 PubMed Central PMC7615308.
- Lyraki, R, Grabek, A, Tison, A, Weerasinghe Arachchige, LC, Peitzsch, M, Bechmann, N et al.. Crosstalk between androgen receptor and WNT/β-catenin signaling causes sex-specific adrenocortical hyperplasia in mice. Dis Model Mech. 2023;16 (6):. doi: 10.1242/dmm.050053. PubMed PMID:37102205 PubMed Central PMC10184674.
- Mayère, C, Regard, V, Perea-Gomez, A, Bunce, C, Neirijnck, Y, Djari, C et al.. Origin, specification and differentiation of a rare supporting-like lineage in the developing mouse gonad. Sci Adv. 2022;8 (21):eabm0972. doi: 10.1126/sciadv.abm0972. PubMed PMID:35613264 PubMed Central PMC10942771.
- Warr, N, Siggers, P, May, J, Chalon, N, Pope, M, Wells, S et al.. Gadd45g is required for timely Sry expression independently of RSPO1 activity. Reproduction. 2022;163 (6):333-340. doi: 10.1530/REP-21-0443. PubMed PMID:35315790 PubMed Central PMC9066659.
- Le Rolle, M, Massa, F, Siggers, P, Turchi, L, Loubat, A, Koo, BK et al.. Arrest of WNT/β-catenin signaling enables the transition from pluripotent to differentiated germ cells in mouse ovaries. Proc Natl Acad Sci U S A. 2021;118 (30):. doi: 10.1073/pnas.2023376118. PubMed PMID:34301885 PubMed Central PMC8325354.
- Mayère, C, Neirijnck, Y, Sararols, P, Rands, CM, Stévant, I, Kühne, F et al.. Single-cell transcriptomics reveal temporal dynamics of critical regulators of germ cell fate during mouse sex determination. FASEB J. 2021;35 (4):e21452. doi: 10.1096/fj.202002420R. PubMed PMID:33749946 .
- Chassot, AA, Le Rolle, M, Jolivet, G, Stevant, I, Guigonis, JM, Da Silva, F et al.. Retinoic acid synthesis by ALDH1A proteins is dispensable for meiosis initiation in the mouse fetal ovary. Sci Adv. 2020;6 (21):eaaz1261. doi: 10.1126/sciadv.aaz1261. PubMed PMID:32494737 PubMed Central PMC7244317.
- Richardson, N, Gillot, I, Gregoire, EP, Youssef, SA, de Rooij, D, de Bruin, A et al.. Sox8 and Sox9 act redundantly for ovarian-to-testicular fate reprogramming in the absence of R-spondin1 in mouse sex reversals. Elife. 2020;9 :. doi: 10.7554/eLife.53972. PubMed PMID:32450947 PubMed Central PMC7250573.
- Tang, F, Richardson, N, Albina, A, Chaboissier, MC, Perea-Gomez, A. Mouse Gonad Development in the Absence of the Pro-Ovary Factor WNT4 and the Pro-Testis Factor SOX9. Cells. 2020;9 (5):. doi: 10.3390/cells9051103. PubMed PMID:32365547 PubMed Central PMC7291083.
- De Cian, MC, Gregoire, EP, Le Rolle, M, Lachambre, S, Mondin, M, Bell, S et al.. R-spondin2 signaling is required for oocyte-driven intercellular communication and follicular growth. Cell Death Differ. 2020;27 (10):2856-2871. doi: 10.1038/s41418-020-0547-7. PubMed PMID:32341451 PubMed Central PMC7493947.
- Vidal, VP, Jian-Motamedi, F, Rekima, S, Gregoire, EP, Szenker-Ravi, E, Leushacke, M et al.. R-spondin signalling is essential for the maintenance and differentiation of mouse nephron progenitors. Elife. 2020;9 :. doi: 10.7554/eLife.53895. PubMed PMID:32324134 PubMed Central PMC7228766.
- Grabek, A, Dolfi, B, Klein, B, Jian-Motamedi, F, Chaboissier, MC, Schedl, A et al.. The Adult Adrenal Cortex Undergoes Rapid Tissue Renewal in a Sex-Specific Manner. Cell Stem Cell. 2019;25 (2):290-296.e2. doi: 10.1016/j.stem.2019.04.012. PubMed PMID:31104943 .
- Stévant, I, Kühne, F, Greenfield, A, Chaboissier, MC, Dermitzakis, ET, Nef, S et al.. Dissecting Cell Lineage Specification and Sex Fate Determination in Gonadal Somatic Cells Using Single-Cell Transcriptomics. Cell Rep. 2019;26 (12):3272-3283.e3. doi: 10.1016/j.celrep.2019.02.069. PubMed PMID:30893600 .
- Gregoire, EP, Stevant, I, Chassot, AA, Martin, L, Lachambre, S, Mondin, M et al.. NRG1 signalling regulates the establishment of Sertoli cell stock in the mouse testis. Mol Cell Endocrinol. 2018;478 :17-31. doi: 10.1016/j.mce.2018.07.004. PubMed PMID:30040984 .
- Chassot, AA, Le Rolle, M, Jourden, M, Taketo, MM, Ghyselinck, NB, Chaboissier, MC et al.. Constitutive WNT/CTNNB1 activation triggers spermatogonial stem cell proliferation and germ cell depletion. Dev Biol. 2017;426 (1):17-27. doi: 10.1016/j.ydbio.2017.04.010. PubMed PMID:28456466 .
- Minkina, A, Lindeman, RE, Gearhart, MD, Chassot, AA, Chaboissier, MC, Ghyselinck, NB et al.. Retinoic acid signaling is dispensable for somatic development and function in the mammalian ovary. Dev Biol. 2017;424 (2):208-220. doi: 10.1016/j.ydbio.2017.02.015. PubMed PMID:28274610 PubMed Central PMC5411265.
- De Cian M.C., Pauper E., Bandiera R., Vidal V.P.I., Sacco S., Gregoire E.P., Chassot A.A., Panzolini C., Wilhelm D., Pailhoux E., Youssef S.A., de Bruin A., Teerds K., Schedl A., Gillot I. and Chaboissier M.C. (2017). Amplification of R-spondin1 signaling induces granulosa cell fate defects and cancers in mouse adult ovary. Oncogene 36(2):208-218. PMID: 27270435
2025 - Équipe labellisée - FRM
2025 - Prix Charles Thibault

These Women Who Are Breaking New Ground: Two iBV Researchers Honored!
Read More

NR2F2: a key player in fetal masculinization
Read More

Marie-Christine Chaboissier awarded the 2025 Charles Thibault Prize !
Read More
Lockdown is not idleness!
Read More
iBV - Institut de Biologie Valrose
"Centre de Biochimie"
Université Nice Sophia Antipolis
Faculté des Sciences
Parc Valrose
06108 Nice cedex 2
