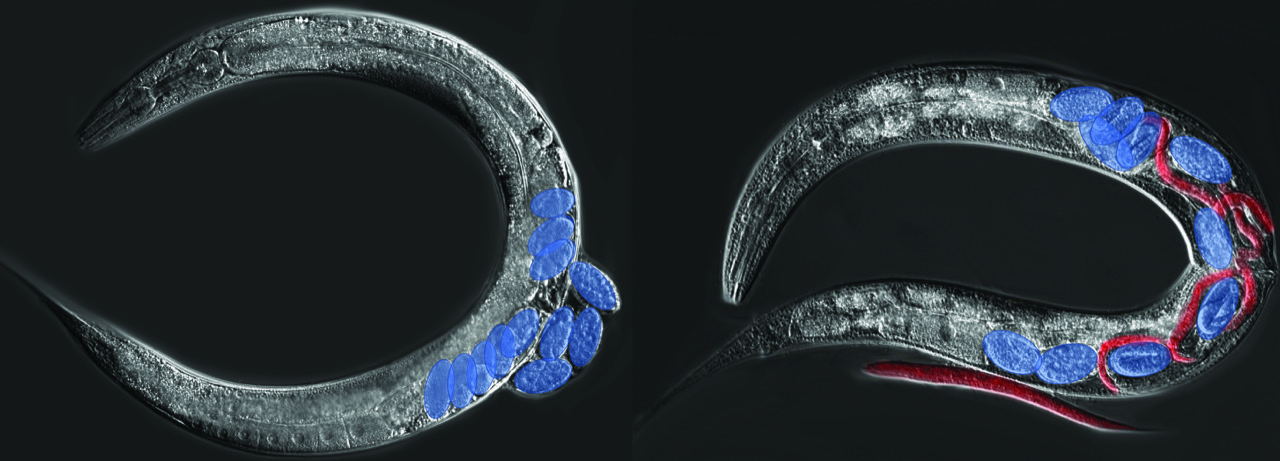
Genetic assimilation – the evolutionary process during which an initially environmentally sensitive phenotype becomes genetically fixed and environmentally insensitive – represents a fundamental but poorly understood concept in evolutionary biology. The nematode Caenorhabditis elegans displays a particularly dramatic example of environmentally-induced phenotypic plasticity: matricidal hatching. In response to starvation and other stressful environments, mothers stop laying eggs, so that larvae will hatch and develop internally, thereby killing their mother. Matricidal hatching is an extreme case of maternal self-sacrifice as internally developing progeny profit from feeding on the decaying mother allowing development and survival in nutrient-scarce environments. The vast majority of natural C. elegans populations display such plastic matricidal hatching, with egg-laying in food-rich conditions and matricidal hatching upon starvation.
In a new study published in Science Advances, the team of Christian Braendle made the surprising finding that certain natural C. elegans isolates exhibit fixed, constitutive matricidal hatching, even in the presence of food. The underlying causative variant is a single amino acid change in the gene kcnl-1, encoding a small-conductance calcium-activated (SK) potassium channel subunit. This gain-of-function mutation (V530L) alters the S6 transmembrane domain of KCNL-1 to cause inappropriate activation of the K+ channel, leading to reduced vulval muscle excitability, and thus reduced expulsion of embryos irrespective of the environment, ultimately leading to matricidal hatching. Using allelic replacement lines, they also showed that this amino acid change is sufficient to induce constitutive matricidal hatching whilst restoring the canonical sequence of the KCNL-1 protein in JU751 abolishes internal hatching, thereby doubling its lifetime reproductive fitness in laboratory food conditions. This KCNL-1 V530L variant was found in multiple wild isolates from different geographic locations, suggesting its apparent maintenance in specific natural contexts. Finally, long-term competition experiments in regimes with fluctuating starvation confirmed that such amino acid variation at the KCNL-1 locus can be maintained.
These results are among the first to show how a single nucleotide change underlies fixation an ancestrally environmentally-induced life history trait.
This work was published in Science Advances and highlighted by INSB (Institut des Sciences Biologiques du CNRS).