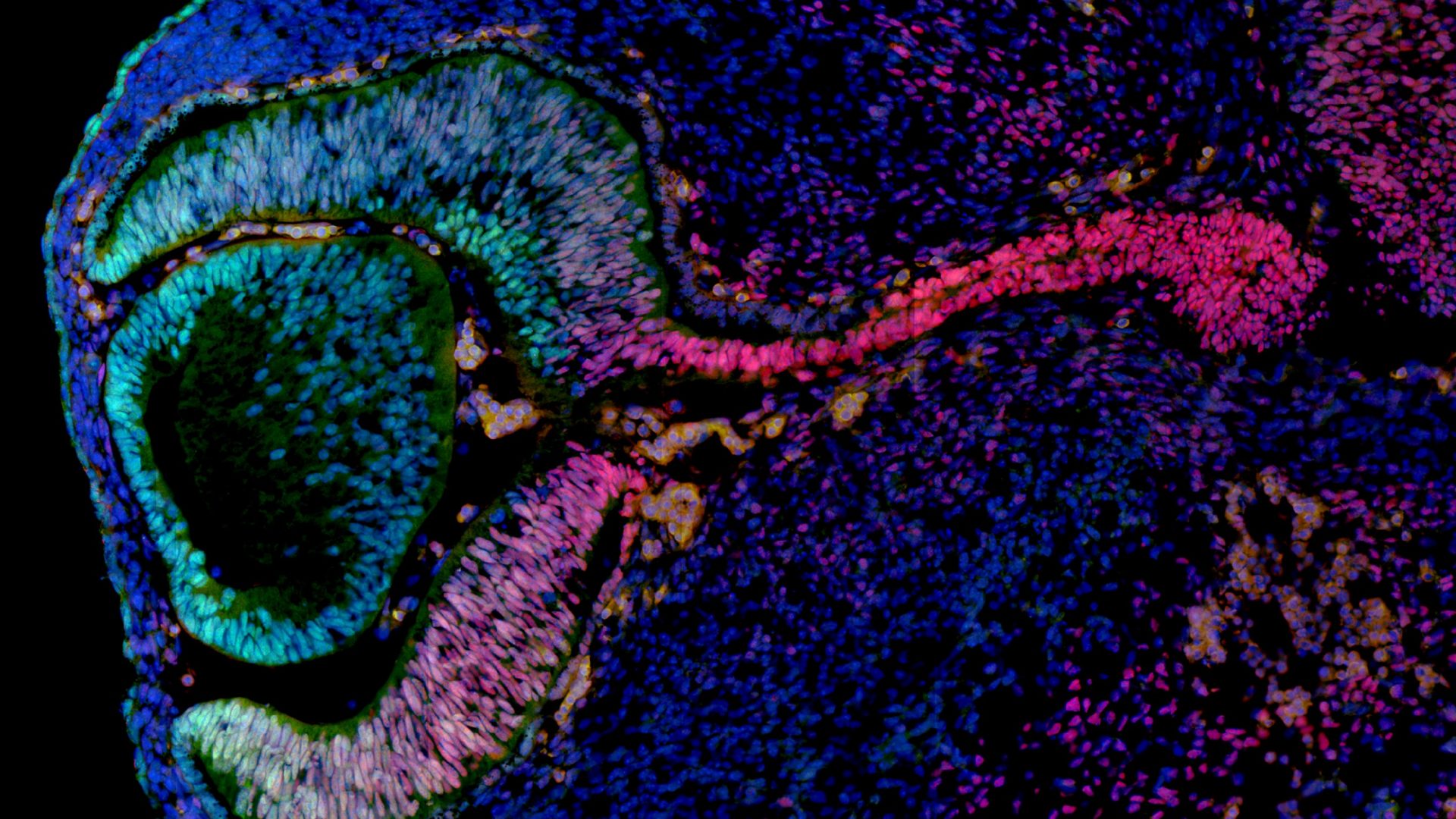
Mouse Nr2f1 haploinsufficiency unveils new pathological mechanisms of a human optic atrophy syndrome.
EMBO Mol Med. 2019 Jul 18:e10291. doi: 10.15252/emmm.201910291. [Epub ahead of print]
Bertacchi M1,2, Gruart A3, Kaimakis P4,5, Allet C6,7, Serra L1,8, Giacobini P6,7, Delgado-García JM3, Bovolenta P4,5, Studer M1.
Author information
1. CNRS, Inserm, iBV, Université Côte d’Azur, Nice, France.
2. Fondazione IRCCS Istituto Neurologico Carlo Besta, Milan, Italy.
3. Division of Neurosciences, Pablo de Olavide University, Seville, Spain.
4. Centro de Biología Molecular “Severo Ochoa”, CSIC-UAM, Madrid, Spain.
5. CIBER de Enfermedades Raras (CIBERER), Campus de la Universidad Autónoma de Madrid, Madrid, Spain.
6. Jean-Pierre Aubert Research Center (JPArc), Laboratory of Development and Plasticity of the Neuroendocrine Brain, UMR-S 1172, Inserm, Lille, France.
7. University of Lille, FHU 1,000 Days for Health, Lille, France.
8. Department of Biotechnology and Biological Sciences, University of Milano-Bicocca, Milano, Italy.
Abstract
Optic nerve atrophy represents the most common form of hereditary optic neuropathies leading to vision impairment. The recently described Bosch-Boonstra-Schaaf optic atrophy (BBSOA) syndrome denotes an autosomal dominant genetic form of neuropathy caused by mutations or deletions in the NR2F1 gene. Herein, we describe a mouse model recapitulating key features of BBSOA patients-optic nerve atrophy, optic disc anomalies, and visual deficits-thus representing the only available mouse model for this syndrome. Notably, Nr2f1-deficient optic nerves develop an imbalance between oligodendrocytes and astrocytes leading to postnatal hypomyelination and astrogliosis. Adult heterozygous mice display a slower optic axonal conduction velocity from the retina to high-order visual centers together with associative visual learning deficits. Importantly, some of these clinical features, such the optic nerve hypomyelination, could be rescued by chemical drug treatment in early postnatal life. Overall, our data shed new insights into the cellular mechanisms of optic nerve atrophy in BBSOA patients and open a promising avenue for future therapeutic approaches.
PMID: 31318166
DOI: 10.15252/emmm.201910291