Researchers at CNRS, Université Côte d’Azur and Inserm have demonstrated a new mechanism related to the onset of migraine. In a protein that can normally inhibit electrical activity, a genetic mutation causes dysfunction that causes migraines. These results, published in Neuron on December 17, 2018, open a new path for the development of anti-migraine medicines.
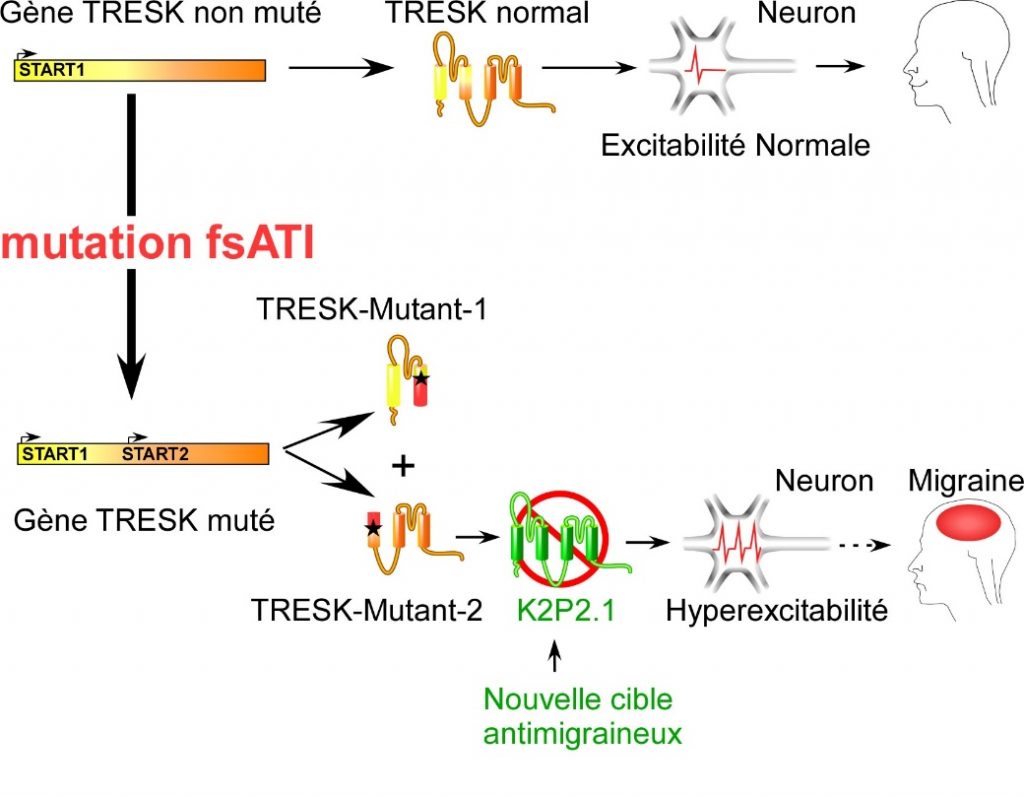
Even though 15% of the adult population worldwide suffers from migraines, no long-term, effective, curative treatment has been marketed to date. Migraine episodes are related, among other factors, to electric hyperexcitability in sensory neurons. Their electrical activity is controlled by proteins that generate current called ion channels, specifically by the TRESK channel, which inhibits electrical activity. The researchers have shown that a mutation in the gene coding for this protein causes a split between two dysfunctional proteins: one is inactive and the other targets other ion channels (K2P2.1), greatly stimulating the electrical activity of the neurons, causing migraines.
Though researchers had already shown the hereditary nature of migraines, they did not know the mechanism. By demonstrating that the TRESK split causes hyperexcitability in sensory neurons and triggers the migraine, this work, carried out at the Institut de Biologie Valrose (CNRS/Inserm/Université Côte d’Azur), is a new research path for the development of anti-migraine medicines. A patent application has been filed1: the scope is targeting K2P2.1 channels to reduce the electrical activity of neurons and prevent migraines from being triggered.
What is more, the researchers propose that this unpublished mechanism, causing the formation of two proteins instead of just one, should now be considered for the study of other diseases related to genetic mutations and for diagnosing them.
1 Patent PCT/EP2018/067581 “Methods and compositions for treating migraine”
Bibliography
Migraine-associated TRESK mutations increase neuronal excitability through alternative translation initiation and inhibition of TREK. Perrine Royal, Alba Andres-Bilbe, Pablo Ávalos Prado, Clément Verkest, Brigitte Wdziekonski, Sébastien Schaub, Anne Baron, Florian Lesage, Xavier Gasull, Joshua Levitz, Guillaume Sandoz, Neuron, December 17, 2018.
https://www.cell.com/neuron/fulltext/S0896-6273(18)31048-1
Contacts
CNRS researcher l Guillaume Sandoz l T +33 4 92 07 68 04 l guillaume.sandoz@unice.fr
Press Book : http://ibv.unice.fr/wp-content/uploads/Retombees-Migraine-2.pdf