Ion channels are essential players in physiology, underlying nerve impulse and the activation of a wide variety of biological processes in many cell types, including heart beating. They are protein complexes made up of an α-pore-forming alpha subunit associated with β-auxiliary regulatory beta subunits. As for other protein complexes, it is well admitted that auxiliary β-subunits are specific to only superfamily of ion channel. KCNE1 is well known example of β-subunit that was thought to associate exclusively with voltage-dependent potassium channels to generate the Iks current whose dysfunction leads to cardiac arrhythmia. But the lab of Guillaume Sandoz at the iBV shifted our perception of protein complexes and ion channel classification. The lab found that KCNE1 subunit can, in addition, serve as an auxiliary subunit of TMEMA16A chloride channels which belongs to another protein superfamily the anoctamin. This came indeed as a big surprise challenging the current dogma, as these two superfamilies of proteins are phylogenetically totally unrelated. To make the story even more important, the team found that clinically relevant inherited KCNE1 mutations, related to cardiac arrhythmia, abolished the ability of KCNE1 to modulate the chloride channel current properties, suggesting a link of this complex with human diseases opening new perspectives for the treatment of cardiac arrhythmias.
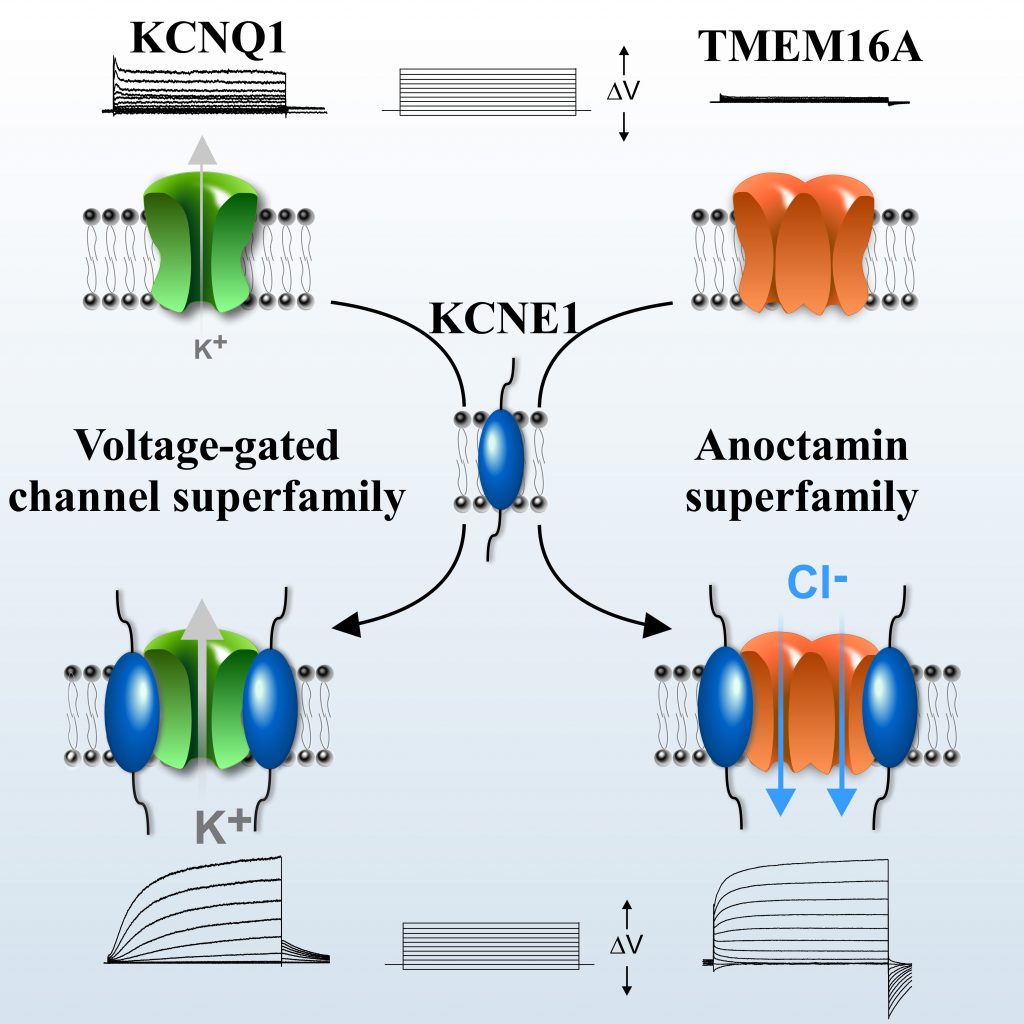
This work was published in Cell and highlighted by INSB (Institut des Sciences Biologiques of CNRS).
In addition, this study allowed to engineer the first designed-TMEM16A agonist which has been patented. This agonist by inducing the cellular secretion of water would be ultimately useful for the treatment of pathologies such as cystic fibrosis or dry eye syndrome1.
1Brevet EP20305795.5