Nature Communications 2023 March 01. doi: 10.1038/s41467-023-36806-4
A photoswitchable inhibitor of TREK channels controls pain in wild-type intact freely moving animals
Arnaud Landra-Willm1,2,3, Ameya Karapurkar4, Alexia Duveau5, Anne Amandine Chassot1,2,3, Lucille Esnault6, Gerard Callejo7,8, Marion Bied1,2,3, Stephanie Häfner1,2,3, Florian Lesage2,3,9, Brigitte Wdziekonski1,2,3, Anne Baron2,3,9, Pascal Fossat5, Laurent Marsollier6, Xavier Gasull7,8, Eric Boué-Grabot5, Michael A. Kienzler10 , Guillaume Sandoz1,2,3
Affiliations
1Université Côte d’Azur, CNRS, INSERM, iBV, Nice, France.
2Laboratories of Excellence, Ion Channel Science and Therapeutics, Nice, France.
3Fédération Hospitalo-Universitaire InovPain, Cote d’Azur University, University Hospital Center, Nice, Provence-Alpes-Côte d’Azur, France.
4University of Maine Department of Chemistry, 178 Munson Rd., Orono, ME 04473, USA.
5Univ. Bordeaux, CNRS, IMN, UMR 5293, F-33000 Bordeaux, France.
6Univ Angers, Nantes Université, INSERM, Immunology and New Concepts in ImmunoTherapy, INCIT, UMR 1302, Angers, France.
7Neurophysiology Laboratory, Department of Biomedicine, Medical School, Institute of Neurosciences, Universitat de Barcelona, c. Casanova 143, 08036 Barcelona, Spain.
8Institut d’Investigacions Biomediques August Pi i Sunyer (IDIBAPS), c. Villarroel 170, 08036 Barcelona, Spain.
9Centre National de la Recherche Scientifique, Institut de Pharmacologie Moléculaire et Cellulaire, Labex ICST, Université Côte d’Azur, INSERM, Valbonne, France.
10University of Connecticut Department of Chemistry, 55 N. Eagleville Rd Storrs, Mansfield, CT 06269, USA.
Abstract
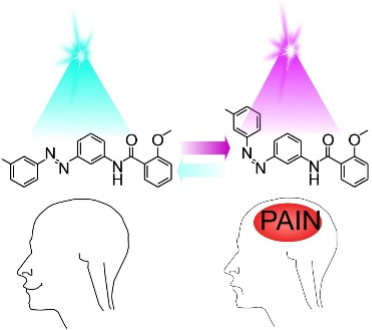
By endowing light control of neuronal activity, optogenetics and photo- pharmacology are powerful methods notably used to probe the transmission of pain signals. However, costs, animal handling and ethical issues have reduced their dissemination and routine use. Here we report LAKI (Light Activated K+ channel Inhibitor), a specific photoswitchable inhibitor of the pain-related two-pore-domain potassium TREK and TRESK channels. In the dark or ambient light, LAKI is inactive. However, alternating transdermal illu- mination at 365 nm and 480 nm reversibly blocks and unblocks TREK/TRESK current in nociceptors, enabling rapid control of pain and nociception in intact and freely moving mice and nematode. These results demonstrate, in vivo, the subcellular localization of TREK/TRESK at the nociceptor free nerve endings in which their acute inhibition is sufficient to induce pain, showing LAKI potential as a valuable tool for TREK/TRESK channel studies. More importantly, LAKI gives the ability to reversibly remote-control pain in a non-invasive and phy- siological manner in naive animals, which has utility in basic and translational pain research but also in in vivo analgesic drug screening and validation, without the need of genetic manipulations or viral infection.
DOI: 10.1038/s41467-023-36806-4

